ALDH2 Deficiency: Causes, Symptoms & Treatment

⏱️ TL:DR ∙ Article in 20s
ALDH2 deficiency is a common genetic condition — especially among East Asian populations — where a mutation prevents properly breaking down acetaldehyde, a toxic metabolite of alcohol. This causes an alcohol flush reaction with facial flushing, nausea, headaches shortly after drinking. More concerning are potential long-term cancer risks from regular alcohol intake over time. While no cure yet exists, responsible drinking and effective supplementation can reduce the risks and symptoms. Emerging research into molecules like alda-1 shows promise for someday improving ALDH2 function. For now, flush prevention pills aid detoxification for more comfortable social drinking.
- ALDH2 enzyme
- ALDH2 deficiency
- Causes of ALDH2 deficiency
- Symptoms of ALDH2 deficiency
- Should I get tested?
- How to get tested
- ALDH2 deficiency treatment
Share this article
Copy and paste this link
Do you turn red in the face or experience uncomfortable symptoms like headaches or nausea after drinking alcohol? If so, you may have a common genetic condition called ALDH2 deficiency. Over a billion people worldwide live with this enzyme deficiency which causes an unpleasant reaction to alcohol.
The good news is that by understanding ALDH2 deficiency, you can learn to manage symptoms and drink more comfortably. In this article, we’ll cover everything from the causes of ALDH2 deficiency and who is affected, to practical solutions like taking “Asian flush” pills. You’ll also learn how alcohol impacts health risks differently for those with this deficiency.
Arm yourself with knowledge so you can take control. Read on as we dive into the science behind ALDH2 deficiency – from what this enzyme does, to symptoms, treatment options, and specific risks to be aware of if you react poorly to drinking.
What Is The ALDH2 Enzyme?
ALDH2 is a very important mitochondrial enzyme found in the liver and mucosa of the upper intestinal tract. But, ALDH2 can also be found in all sorts of other tissues throughout the human body. It plays a key role in breaking down aldehydes. As such, some people refer to this enzyme as mitochondrial aldehyde dehydrogenase.
Those with normal, functioning mitochondrial aldehyde dehydrogenase are able to break down alcohol without issue. However, those who suffer from a deficiency in this enzyme are prone to issues we mentioned at the outlook - red skin, headaches, nausea, etc. That is because this mitochondrial aldehyde dehydrogenase enzyme is incapable of eliminating toxins from the body in a timely manner.
Let's take a deeper look at what mitochondrial aldehyde dehydrogenase deficiency looks like.
What is ALDH2 deficiency?
ALDH2 deficiency refers to a deficiency in a detoxifying enzyme that makes it harder for the liver to break down some toxins that enter the body.
The typical human liver contains two major aldehyde dehydrogenase enzymes, a cytosolic ALDH1 component, and a mitochondrial ALDH2 component.
Many people, notably about 30% to 40% of East Asian Orientals, have only the ALDH1 enzyme and are deficient in the ALDH2 enzyme.
Being deficient in the ALDH2 enzyme means that your body is less able to safely deal with a particular set of toxins that can enter from the external environment.
One example is the toxin acetaldehyde that enters our body when we consume alcoholic beverages. The ALDH2 enzyme is responsible for detoxifying acetaldehyde, so if you're ALDH2 deficient your body will undergo acetaldehyde toxicity whenever you drink alcohol.
This is commonly referred to as alcohol flush reaction or Asian flush (because of the large proportion of Asian people affected).
What causes ALDH2 deficiency?
ALDH2 deficiency is a genetic condition thought to be originally caused by the emergence of rice cultivation in Asia between 7,000 to 10,000 years ago.

So what exactly causes a deficiency in mitochondrial aldehyde dehydrogenase deficiency? You now know that this deficiency is more prominent in Asian individuals, but why?
A mutation in the ALDH2 gene is thought to be the cause, in that it encodes a form of the aldehyde dehydrogenase 2 protein that is very bad at metabolizing environmental toxins like acetaldehyde from alcohol.
A team of researchers headed by Bing Su, a geneticist at the Kunming Institute of Zoology, conducted a study to identify the source of this genetic ALDH2 deficiency.
The team searched for ALDH2 deficiency in 2,275 people across China representing 38 different ethnic groups. They reported that 99% of the people in the southeast region of China had the problem, with less prevalence seen in western China and Tibet.
Notably, the team found a strong geographical correlation between regions with a high prevalence of this issue and archaeological sites in China where rice had been domestically cultivated thousands of years ago. The geographical correlation suggests the potential of a culture-wide dietary catalyst for the gene mutation that causes ALDH2 deficiency.
What are the ALDH2 deficiency symptoms?
The ALDH2 deficiency symptoms can vary depending on the one's sensitivity to the environmental toxin not being broken down.
In the case of alcohol, the specific toxin not being broken down in ALDH2 deficient individuals is acetaldehyde. Improper alcohol metabolism leads to a whole host of issues.
When acetaldehyde enters the body it provokes a histamine release that can cause any of the following short-term ALDH2 deficiency symptoms:
- Erythema - redness of the skin caused by increased blood flow to superficial capillaries in the face, neck and upper body.
- Tachycardia - a heart rate that exceeds the normal resting rate.
- Nausea - uneasy feeling in the upper stomach with an urge to vomit.
- Headache - pain in the head often throbbing or pulsing.
This normally occurs 20 to 30 minutes after consuming a sufficient quantity of alcohol and can take up to 2 to 3 hours to subside depending on the individual partaking in alcohol drinking.
While the symptoms listed above are uncomfortable and at times dangerous, there are also more serious issues associated with alcohol consumption in ALDH2 deficient individuals. In fact, this condition may be a yet unrecognized risk factor for certain cancers.
Longer-term effects of consistent acetaldehyde exposure include an increased risk of various types of cancers: including esophageal cancer risk, squamous cell carcinoma risk. But, there is also evidence suggesting this deficiency contributes to an elevated risk of stroke, liver cirrhosis, and other issues with the esophagus and upper digestive tract.
For more information about common long-term risks facing people with ALDH2 deficiency check out our article titled: Debunking The Asian Flush Cancer Risk.
Should I Get Tested for ALDH2 Deficiency?
At best, ALDH2 deficiency is uncomfortable, unsightly, and embarrassing. At worst, it can be life threatening. We say all this not to scare you, but rather to make you aware of the very real risks that come with alcohol consumption when you're deficient in this enzyme. So, should you get tested?
The easiest way to test for ALDH2 deficiency is to check if you suffer from the symptoms mentioned above. But after receiving a few emails from customers, we looked into more definitive methods of checking for mutations in people's genes.
If you finally want to know whether you have this deficiency (commonly referred to as Asian Flush or Asian Glow) there are a few at-home gene tests that you can complete.
This will tell you once and for all if you have this enzyme deficiency, or if your reaction is actually caused by something else. If you really want to know, you will need to buy and complete a genetic test.
However, for most people, experiencing negative symptoms from alcohol is enough to convince them of their ALDH2 deficiency, even if this reaction may be something else, like an allergy to alcohol
How can I get tested for ALDH2 Deficiency?
Getting tested for ALDH2 deficiency is actually way easier than you'd ever imagine. There are a few reputable companies that will analyze your DNA, and send you back the results.
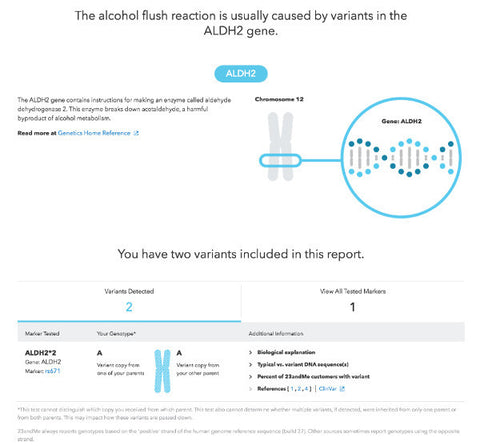
23andMe
The best method we came across is a simple DNA test from the internationally known company 23andMe. From their 'how it works' page:
23andMe is a DNA analysis service providing information and tools for individuals to learn about and explore their DNA.
Quite simply, you send in a saliva sample and they run a full genome analysis on you, uploading the results online.
The test results are (of course!) fascinating, as you will no-doubt found out new things about yourself, and your ancestry that you did not know before.
For the purpose of testing for ALDH2 deficiency, however, we are mostly interested in the part of the report that addresses the ALDH2 gene and alcohol flush reactions.
View a sample of the ALDH2 gene report section here, or sign up via their website to get a full DNA check (we are not affiliated with 23andMe.
ORIG3N
A company called ORIG3N offers specific ALDH2 Deficiency tests for $29. This is currently one of the cheapest ways to get this test completed, without having to also test for everything else (like other common DNA tests).
This test will definitively let you know if you have ineffective ALDH2 genes once and for all.
ORIG3N is a company based in Boston, USA and focus primarily on very specific genetic tests for a cheaper price.
They sell tests designed for metabolism, lactose intolerance, skin health and appearance, nutrition and fitness among many others.
This can be really helpful if you want to test for one thing (Asian Flush) without having to pay to test everything else as well.
Determining whether you really need to get tested for ALDH2 deficiency
These tests are easy, but they aren't free. And, some people have their hesitations about sharing their DNA with a huge corporation - and rightfully so!
If you’re really not sure what to do next, it’s best to speak with your doctor. Negative symptoms such as facial flushing can be a real sign from your body that something is wrong.
Your doctor will be able to give you more information about your specific case, what to do next and whether an ALDH2 deficiency test is worth your time and money.
Consuming alcohol should always be done responsibly and in moderation, especially if you deal with an ALDH2 deficiency.
ALDH2 deficiency treatment
After realizing you may be forced to live with this condition, you may wonder about ALDH2 deficiency treatment.
Preliminary research into treating the enzyme issue is ongoing. To date, the most effective treatment for ALDH2 deficiency is detoxification assistance.
Researchers at Stanford University conducted a study that found that a small molecule called alda-89 could increase the ALDH2 enzyme function in ALDH2 deficient mice.
It does this by causing other enzymes to mimic the function of the broken ALDH2 enzyme.
Unfortunately, alda-89 is not safe for human consumption due to its toxicity, but researchers at Standford are hopeful of finding a safer molecule that performs the same function.
Another molecule that has garnered the attention of researchers is ALDA-1. ALDA-1 has been shown in studies to modulate the kinetic properties of the ALDH2 enzyme and increase its function dramatically.
Although not thoroughly tested for safety in human subjects, research is ongoing to determine the therapeutic benefit of alda-1 in treating ALDH2 deficiency.
Saadeh Suleiman of the Bristol Heart Institute, comments that the research is initially exciting but also cautions that human ALDH2 might not be as easy to work with as it is in rats.
So what is the cure for ALDH2 deficiency?
Unfortunately, there is no cure for this issue, it's just about ALDH2 deficiency treatment and management. Of course, the quick fix is to limit alcohol intake. If you know you have an increased risk of head and neck cancer, and other alcohol-related cancers or diseases, the right thing to do is to avoid excessive alcohol consumption.
But does that mean that you have to abstain from drinking alcohol altogether? Not necessarily! You can still safely indulge in drinking alcohol every now and then without worry - if you follow the recommendations below.
To date, the best way to treatment method has been to address the toxicity issue rather than the enzyme deficiency. This method will vary depending on the particular toxin not being broken down in ALDH2 deficient individuals.
As mentioned above, in the case of alcohol the toxin in question is acetaldehyde.
Research has shown that precise supplementation of a combination of compounds can reduce acetaldehyde toxicity and aid the deficient ALDH2 enzyme in metabolizing alcohol — treating the symptoms immediately, rather than preventing them completely.
To find out more check out our article titled: The Science of an Alcohol Red Face and How to Cure it.
Final Thoughts On ALDH2 Deficiency Causes, Symptoms, & Treatment
Well, there you have it - everything you need to know about aldehyde dehydrogenase deficiency, also known as ALDH2 deficiency. We've covered what exactly this enzyme is and why it is so important, and what causes certain individuals to be deficient in it.
We then covered what symptoms you should look for, and what the risks are for those who have an alcohol drinking habit along with a deficiency in the necessary enzymes for breaking toxic acetaldehyde down. If you believe you could be suffering from this deficiency, we highly encourage you to get tested to find out once and for all.
After that, you'll be able to come up with a plan on how to live with the deficiency. You obviously should curb excessive alcohol abuse, but it doesn't mean you must abstain entirely.
You just need to help your body with breaking down those harmful compounds - and that is where Sunset Alcohol Flush Pills come in. These are your key to continuing to live a comfortable social life without worrying about the effects of ALDH2 deficiency - take a look and see if this is the right treatment for you!
Enjoy drinking again and get Sunset Alcohol Flush Support for
33% off while stocks last!
What’s inside?
We use a pharmacist-formulated blend of Glutathione, Dihydromyricetin, Cysteine, L-Theanine, & B Vitamins to stop alcohol flushing before it can begin.
Learn more
94% of people who try Sunset are satisfied with the results.